;)
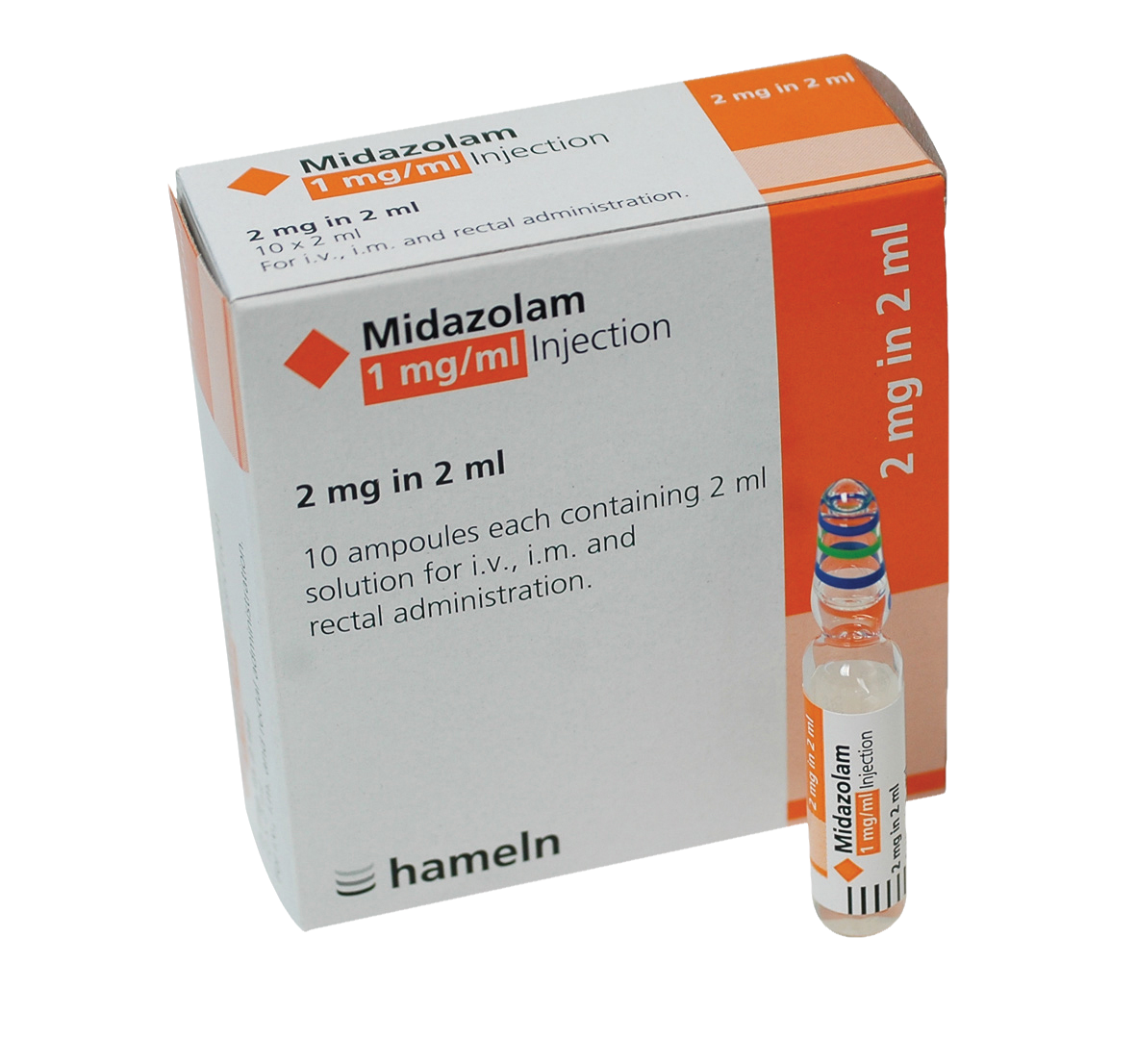
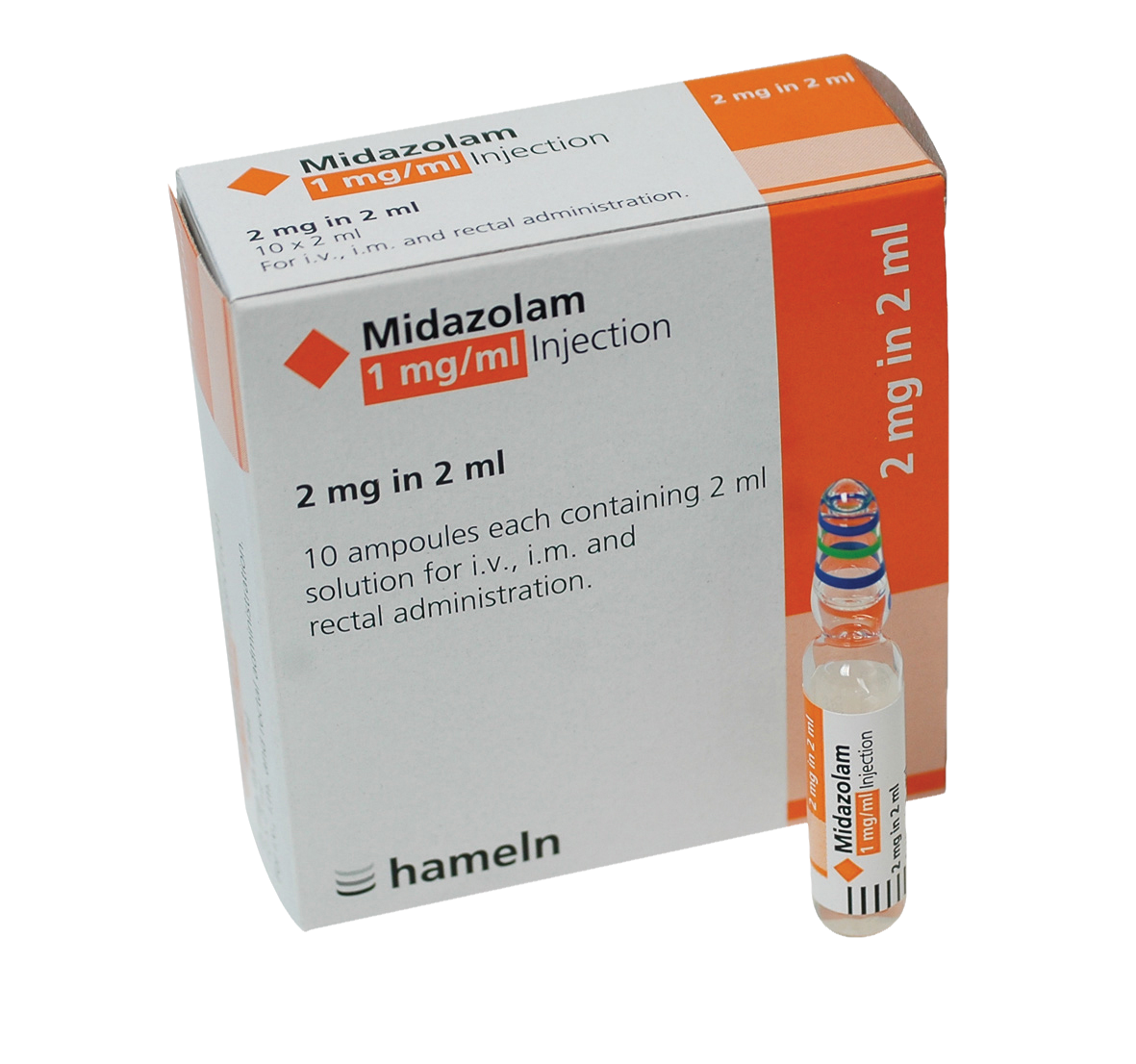
Midazolam 1mg/1ml Solution for Injection/Infustion 2mg in 2ml Ampoule 10pk
EA
Please note that this controlled drug cannot be ordered via our website. To place an order, a completed Controlled Drugs Requisition form (FP10CDF) is required to purchase this item. Click here to download the official form. The form should be printed, completed and signed by an approved healthcare professional, and then posted to Henry Schein UK Holdings Limited, Medcare House, Centurion Close, Gillingham, ME8 0SB. UK WDA(H) 50837. For further information, emergency procedures and support, contact our Sales team on 0800 023 2558. Please note, email requests cannot be accepted.
Restricted item. To purchase, you will need to be a dentist or doctor with a valid GDC/GMC or equivalent licence.
Prescription-only medicine. Must be prescribed by a qualified health professional.
This product does not have a long shelf life. For more information, please contact our Customer Service team via email at ukcustomerservices@henryschein.co.uk or by phoning 0800 028 1533.
Non-returnable unless faulty.
Description
Product Attributes
Return Policy
Specifications
Manufacturer
Hameln Pharmaceuticals
GDC/GMC
Required
Manufacturer Product Code
112-8529
NHS VMPP Code
4752211000001105
Pack Qty
10
Product Sub Type
IV Drugs
Product Type
Pharmaceuticals